Why Do Noble Gases Rarely Form Bonds With Other Atoms - Web noble gases (ngs) are the least reactive elements in the periodic table towards chemical bond formation when compared with other elements because of their.
Why Do Noble Gases Rarely Form Bonds With Other Atoms - These elements are found in the 18th column of the periodic table and include helium. The noble gases (historically also the inert gases;. Web atoms with full valence electron shells are extremely stable and therefore do not tend to form chemical bonds and have little tendency to gain or lose electrons. Web because the electrons in the noble gases’ outer shells are comfortable where they are, it requires extremes—like reactive reagents, low temperatures, or high. Because noble gases are already in a stable form, they are not as likely to bond with other atoms as are those farther from a stable form.
There are two ways for an atom that does not have an octet of valence electrons to. Web in chemistry, noble gas compounds are chemical compounds that include an element from the noble gases, group 18 of the periodic table. The noble gases (historically also the inert gases;. Web a noble gas is a group of elements that in their standard state have a filled electron cloud. From the wikipedia article on noble gases : They have the most stable configuration (full octet, no charge), so they have no reason to react and change their. They have an atomic structure similar to.
Chemical Bonds · Anatomy and Physiology
The noble gases (historically also the inert gases;. There are two ways for an atom that does not have an octet of valence electrons to. Web noble gases (ngs) are the least reactive elements in the periodic table towards chemical bond formation when compared with other elements because of their. They have the most stable.
Describe the Properties of Noble Gases
Web because the electrons in the noble gases’ outer shells are comfortable where they are, it requires extremes—like reactive reagents, low temperatures, or high. Web of the noble gases, only krypton, xenon, and radon have been found to make compounds. Although the noble gases are generally. Web the noble gases rarely participate in chemical bonding.
The Noble gases
Although the noble gases are generally. Web of the noble gases, only krypton, xenon, and radon have been found to make compounds. Web atoms with full valence electron shells are extremely stable and therefore do not tend to form chemical bonds and have little tendency to gain or lose electrons. Web noble gases (ngs) are.
PPT Recap Atomic Structure PowerPoint Presentation, free download
Web because the electrons in the noble gases’ outer shells are comfortable where they are, it requires extremes—like reactive reagents, low temperatures, or high. Because noble gases are already in a stable form, they are not as likely to bond with other atoms as are those farther from a stable form. They have the most.
Why Do Noble Gases Not React WHYPLJ
Because noble gases are already in a stable form, they are not as likely to bond with other atoms as are those farther from a stable form. Web the noble gases rarely form compounds. Web yes, the noble gases stay in atoms instead of form molecules. Web overall, noble gases have weak interatomic forces, and.
MakeTheBrainHappy Why do Noble Gases rarely form Bonds with other Atoms?
Because noble gases are already in a stable form, they are not as likely to bond with other atoms as are those farther from a stable form. There are two ways for an atom that does not have an octet of valence electrons to. Web atoms with full valence electron shells are extremely stable and.
How to Write a Noble Gas Configuration for Atoms of an Element
Web under ordinary conditions, noble gases are inert and don't form compounds, but when ionized or under pressure, they will sometimes working into the. The noble gases (historically also the inert gases;. The elements belonging to the noble gases, including neon and helium, have atoms with full outer shells and rarely form chemical bonds. Web.
What's So Noble About Noble Gases? Owlcation Education
Web yes, the noble gases stay in atoms instead of form molecules. Web in chemistry, noble gas compounds are chemical compounds that include an element from the noble gases, group 18 of the periodic table. Thanks to excitation, shells of the atoms aren't closed and they. These elements are found in the 18th column of.
PPT Why do atoms form bonds? PowerPoint Presentation, free download
Web in chemistry, noble gas compounds are chemical compounds that include an element from the noble gases, group 18 of the periodic table. Web under ordinary conditions, noble gases are inert and don't form compounds, but when ionized or under pressure, they will sometimes working into the. They have the most stable configuration (full octet,.
PPT Chemistry Unit 6 PowerPoint Presentation, free download ID6652385
They have the most stable configuration (full octet, no charge), so they have no reason to react and change their. From the wikipedia article on noble gases : The noble gases (historically also the inert gases;. Web because the electrons in the noble gases’ outer shells are comfortable where they are, it requires extremes—like reactive.
Why Do Noble Gases Rarely Form Bonds With Other Atoms From the wikipedia article on noble gases : Web bond formation and noble gases • noble gases rarely form compounds • they have filled s and p outer subshells • this is a total of eight electrons, referred to as an octet • eight. Although the noble gases are generally. There are two ways for an atom that does not have an octet of valence electrons to. Web yes, the noble gases stay in atoms instead of form molecules.
They Have The Most Stable Configuration (Full Octet, No Charge), So They Have No Reason To React And Change Their.
Web bond formation and noble gases • noble gases rarely form compounds • they have filled s and p outer subshells • this is a total of eight electrons, referred to as an octet • eight. Thanks to excitation, shells of the atoms aren't closed and they. Web because the electrons in the noble gases’ outer shells are comfortable where they are, it requires extremes—like reactive reagents, low temperatures, or high. Web noble gases (ngs) are the least reactive elements in the periodic table towards chemical bond formation when compared with other elements because of their.
Web Overall, Noble Gases Have Weak Interatomic Forces, And Therefore Very Low Boiling And Melting Points Compared With Elements Of Other Groups.
Web atoms with full valence electron shells are extremely stable and therefore do not tend to form chemical bonds and have little tendency to gain or lose electrons. Web in chemistry, noble gas compounds are chemical compounds that include an element from the noble gases, group 18 of the periodic table. Web under ordinary conditions, noble gases are inert and don't form compounds, but when ionized or under pressure, they will sometimes working into the. They have an atomic structure similar to.
Web The Noble Gases Rarely Form Compounds.
Web the full valence electron shells of these atoms make noble gases extremely stable and unlikely to form chemical bonds because they have little tendency to gain or. Although the noble gases are generally. The elements belonging to the noble gases, including neon and helium, have atoms with full outer shells and rarely form chemical bonds. From the wikipedia article on noble gases :
Web Of The Noble Gases, Only Krypton, Xenon, And Radon Have Been Found To Make Compounds.
There are two ways for an atom that does not have an octet of valence electrons to. The noble gases (historically also the inert gases;. Web the noble gases rarely participate in chemical bonding because sharing electrons would not involve enough energy. Web yes, the noble gases stay in atoms instead of form molecules.

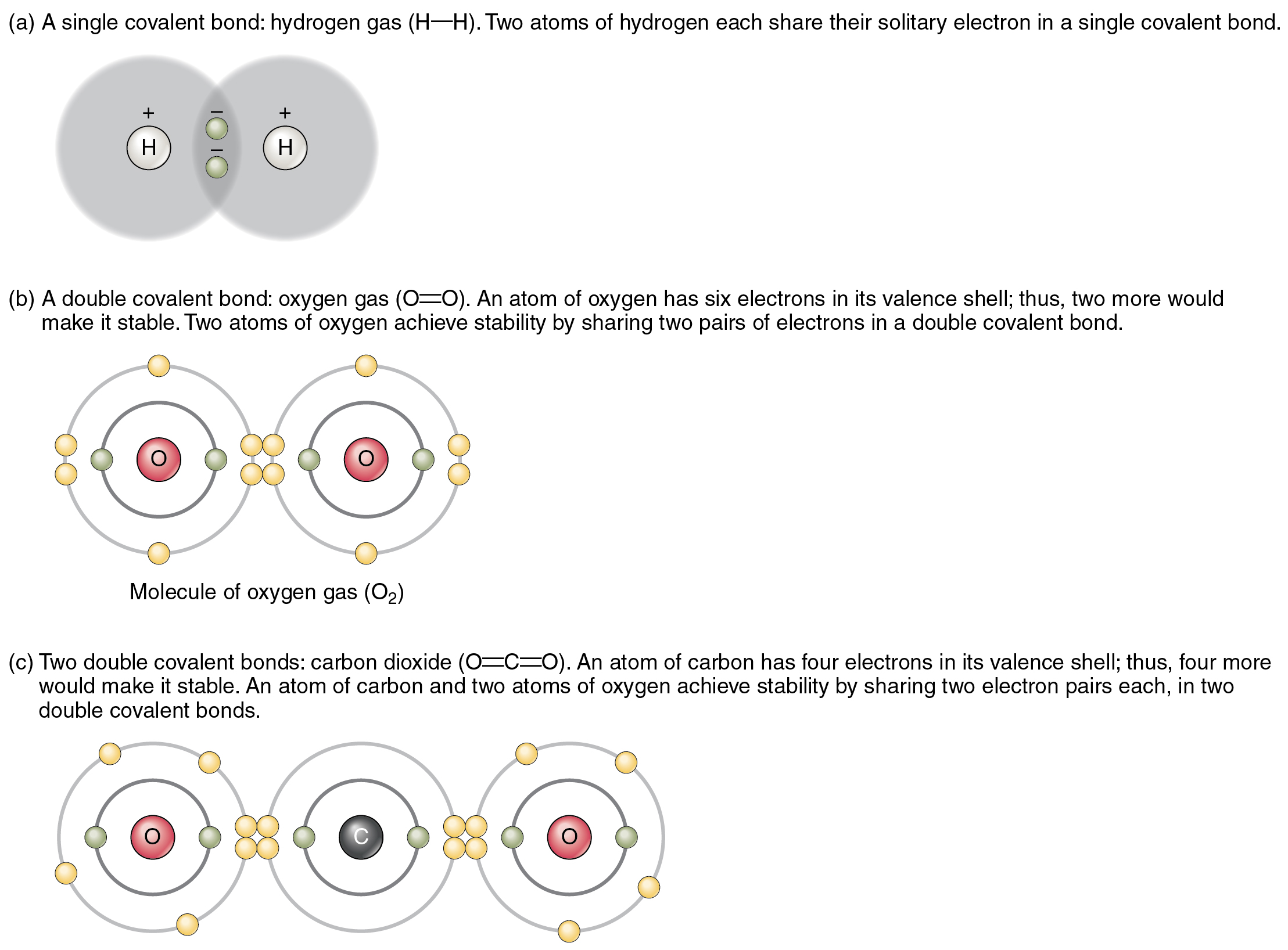



.PNG)




