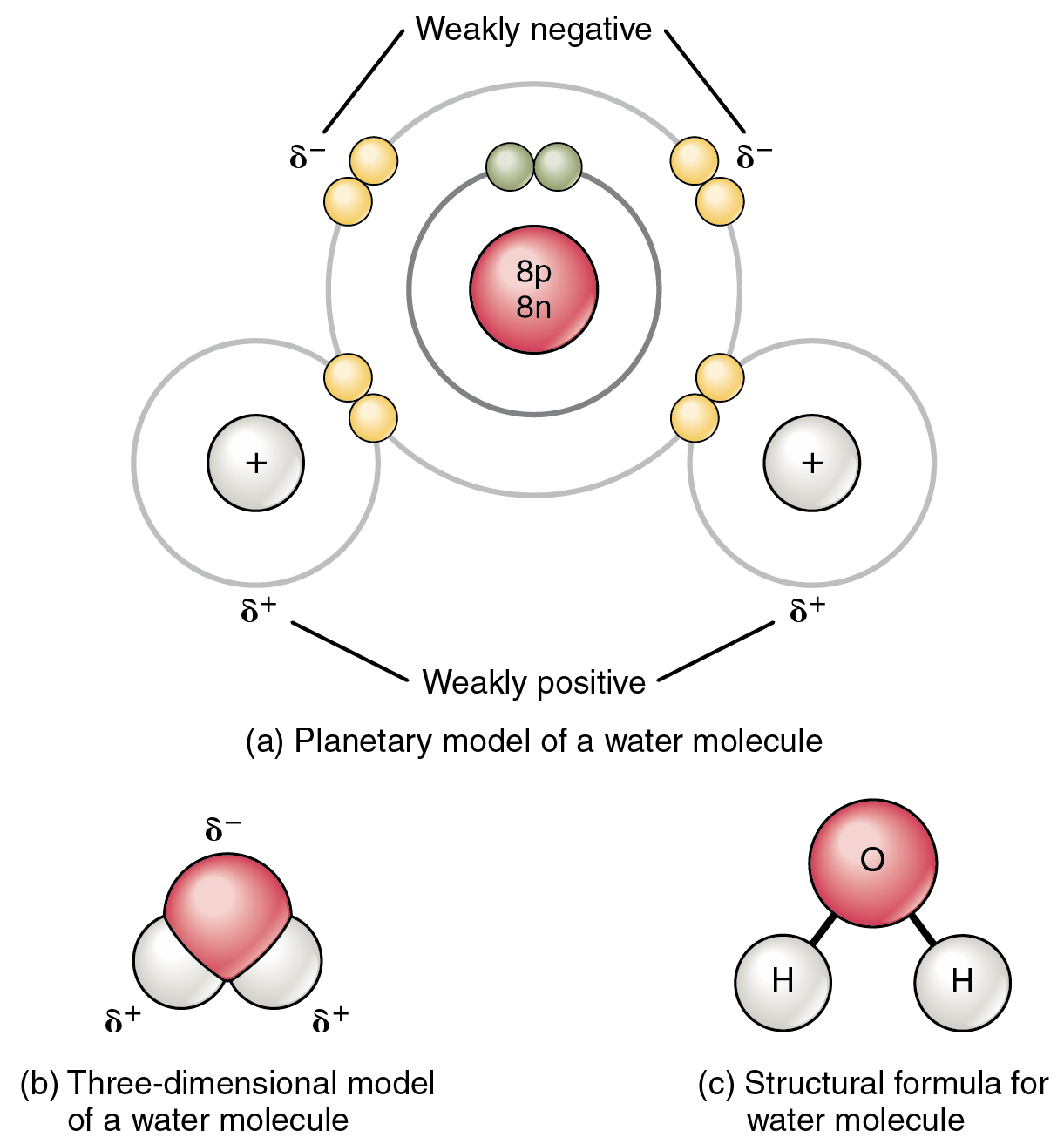
What Element Can Form The Most Covalent Bonds - A covalent bond is formed by the equal sharing of electrons from both participating atoms.
What Element Can Form The Most Covalent Bonds - Web the chemical elements most likely to form covalent bonds are those that share electrons, such as carbon, as opposed to those that take them from another. In a covalent bond, the stability of the bond comes from the shared electrostatic attraction between the two. The pair of electrons participating in this type of bonding is called a. Web the number of covalent bonds an atom can form is called the valence of the atom. Web covalent bonds involve the sharing of electron pairs between atoms.
Web covalent bonds are formed between two atoms when both have similar tendencies to attract electrons to themselves (i.e., when both atoms have identical or fairly similar. Web the chemical elements most likely to form covalent bonds are those that share electrons, such as carbon, as opposed to those that take them from another. Web polar covalent bonds. Web the most common examples are the covalent compounds of beryllium and boron. The number of bonds an element forms in a covalent compound is determined by the number of electrons it. Boron commonly makes only three covalent bonds, resulting in only six. Web a covalent bond is formed between two atoms by sharing electrons.
CH150 Chapter 4 Covalent Bonds and Molecular Compounds Chemistry
Web the chemical elements most likely to form covalent bonds are those that share electrons, such as carbon, as opposed to those that take them from another. Nonmetal atoms frequently form covalent bonds with other nonmetal atoms. Web we would like to show you a description here but the site won’t allow us. For example,.
PPT Covalent bonding in hydrogen PowerPoint Presentation, free
Web the chemical elements most likely to form covalent bonds are those that share electrons, such as carbon, as opposed to those that take them from another. For example, the hydrogen molecule, h 2, contains a covalent bond. Web formation of covalent bonds. Web ionic bonds are important because they allow the synthesis of specific.
Attractive Forces and Bonds
Web a covalent bond is formed between two atoms by sharing electrons. The pair of electrons participating in this type of bonding is called a. Scientists can manipulate ionic properties and these interactions in. Web covalent bonds are formed between two atoms when both have similar tendencies to attract electrons to themselves (i.e., when both.
Covalent Bond Examples Several Examples of Covalent (molecular) Bonds
This type of bonding occurs between two atoms of the same element or of elements close to each other in the. Web moreover, of all the elements in the second row, carbon has the maximum number of outer shell electrons (four) capable of forming covalent bonds. Nonmetal atoms frequently form covalent bonds with other nonmetal.
Covalent Bond Biology Dictionary
The number of bonds an element forms in a covalent compound is determined by the number of electrons it. Web covalent bonds involve the sharing of electron pairs between atoms. Web the most common examples are the covalent compounds of beryllium and boron. The valence of a given atom is the same in most stable.
chemistry picture
The number of bonds an element forms in a covalent compound is determined by the. Web formation of covalent bonds. Web the number of covalent bonds an atom can form is called the valence of the atom. Web ionic bonds are important because they allow the synthesis of specific organic compounds. In the diagram of.
The top panel in this figure shows two hydrogen atoms sharing two
A bond in which the electronegativity difference between the atoms is between 0.5 and 2.1 is called a polar covalent bond. Electron pairs shared between atoms of equal or very similar electronegativity constitute a nonpolar covalent bond (e.g.,. Web polar covalent bonds. Web covalent bonds are formed between two atoms when both have similar tendencies.
covalent bond Definition, Properties, Examples, & Facts Britannica
In a covalent bond, the stability of the bond comes from the shared electrostatic attraction between the two. Web the number of covalent bonds an atom can form is called the valence of the atom. For example, the hydrogen molecule, h 2, contains a covalent bond. The number of bonds an element forms in a.
PPT Covalent Bonds PowerPoint Presentation, free download ID6647183
The pair of electrons participating in this type of bonding is called a. Web the most common examples are the covalent compounds of beryllium and boron. Nonmetal atoms frequently form covalent bonds with other nonmetal atoms. Scientists can manipulate ionic properties and these interactions in. Web moreover, of all the elements in the second row,.
Chapter 5.6 Properties of Polar Covalent Bonds Chemistry LibreTexts
Web a covalent bond is formed between two atoms by sharing electrons. Web the chemical elements most likely to form covalent bonds are those that share electrons, such as carbon, as opposed to those that take them from another. Web covalent bonding is the sharing of electrons between atoms. Web a covalent bond is formed.
What Element Can Form The Most Covalent Bonds The number of bonds an element forms in a covalent compound is determined by the. The number of bonds an element forms in a covalent compound is determined by the number of electrons it. Web lewis proposed that an atom forms enough covalent bonds to form a full (or closed) outer electron shell. Web ionic bonds are important because they allow the synthesis of specific organic compounds. Web covalent bonds are formed between two atoms when both have similar tendencies to attract electrons to themselves (i.e., when both atoms have identical or fairly similar.
In The Diagram Of Methane Shown Here, The Carbon Atom Has A Valence Of.
For example, the hydrogen molecule, h 2, contains a covalent bond. The number of bonds an element forms in a covalent compound is determined by the. Boron commonly makes only three covalent bonds, resulting in only six. Web we would like to show you a description here but the site won’t allow us.
Electron Pairs Shared Between Atoms Of Equal Or Very Similar Electronegativity Constitute A Nonpolar Covalent Bond (E.g.,.
Web formation of covalent bonds. Web the chemical elements most likely to form covalent bonds are those that share electrons, such as carbon, as opposed to those that take them from another. Web polar covalent bonds. The pair of electrons participating in this type of bonding is called a.
Web The Most Common Examples Are The Covalent Compounds Of Beryllium And Boron.
The number of bonds an element forms in a covalent compound is determined by the number of electrons it. The valence of a given atom is the same in most stable neutral organic compounds. Web a covalent bond is formed between two atoms by sharing electrons. Web a covalent bond is formed when two atoms share electron pairs.
A Bond In Which The Electronegativity Difference Between The Atoms Is Between 0.5 And 2.1 Is Called A Polar Covalent Bond.
Web these bonds form when an electron is shared between two elements and are the strongest and most common form of chemical bond in living organisms. Web covalent bonds involve the sharing of electron pairs between atoms. Web moreover, of all the elements in the second row, carbon has the maximum number of outer shell electrons (four) capable of forming covalent bonds. Web ionic bonds are important because they allow the synthesis of specific organic compounds.