How Many Covalent Bonds Can Sulfur Form - Web each of the 3 chlorines then forms a covalent bond by merging the atomic orbital containing its unpaired electron with one of the phosphorus's unpaired electrons.
How Many Covalent Bonds Can Sulfur Form - Web now sulfur has 6 unpaired electrons which means it can form 6 covalent bonds to give a total of 12 electrons around its valence shell. Group 6a form 2 bonds; Group 5a form 3 bonds; Is sulfur trioxide ionic or covalent? Because hydrogen only needs two electrons to fill its valence shell, it is an exception to the octet rule and only.
The potential energy of two separate hydrogen atoms (right) decreases as. Web study with quizlet and memorize flashcards containing terms like if sulfur has an atomic number of 16, how many covalent bonds can it form with other atoms? In fact, it is a multivalent atom, meaning it can make different number of bonds, up to 6, depending on the chemical formula. Sulfur is an essential component of all living cells. Web covalent radius half of the distance between two atoms within a single covalent bond. Which of the following constitutes a covalent. Is sulfur trioxide ionic or covalent?
Question 51110 Socratic
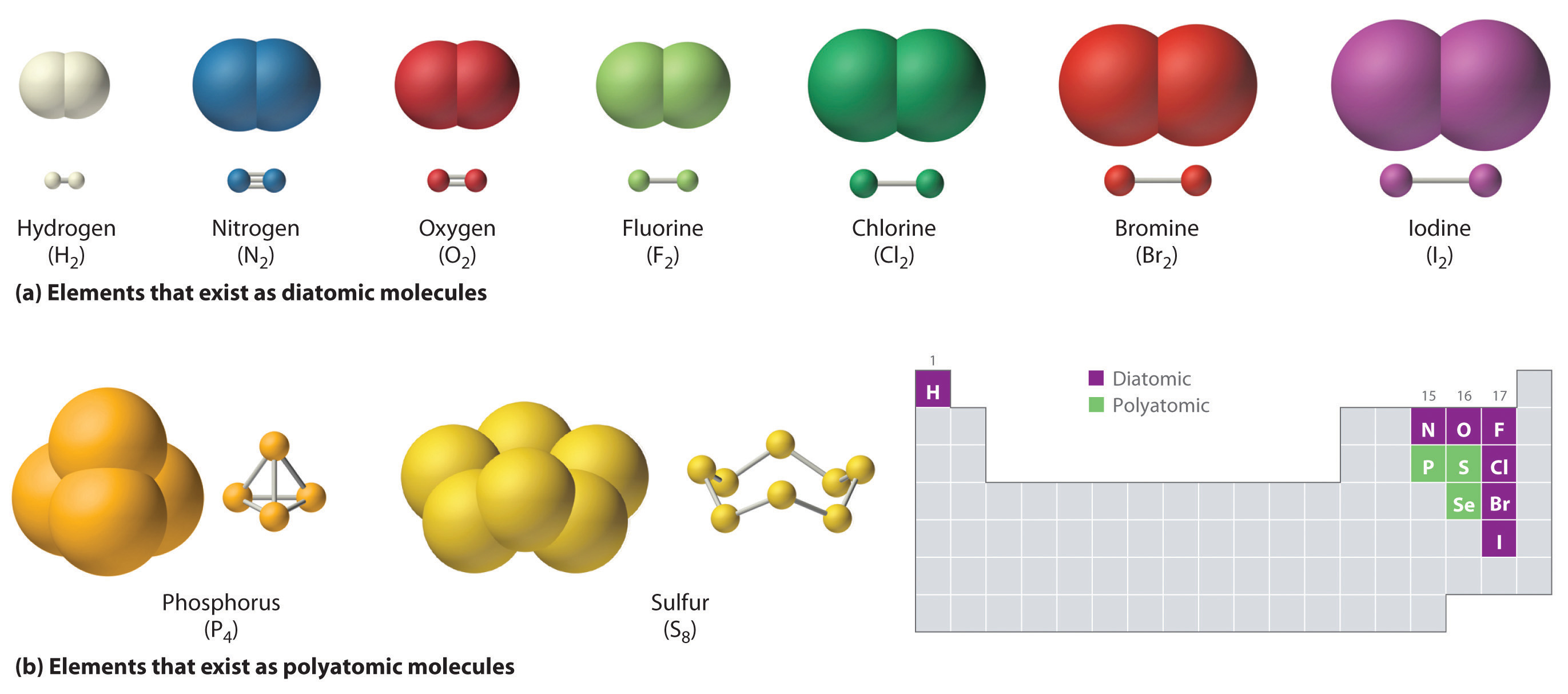
Web how many single covalent bonds can halogens form? Web atoms of different elements will form either one, two, three or four covalent bonds with other atoms. A 70 kg (150 lb) human body contains about 140 grams of sulfur. Web now sulfur has 6 unpaired electrons which means it can form 6 covalent bonds.
covalent bond Definition, Properties, Examples, & Facts Britannica
Web up to 6% cash back how many single covalent bonds would the element sulfur be expected to form in order to obey the octet rule? There is a quick way to work out how many covalent bonds an element. Nonmetal atoms frequently form covalent bonds with other nonmetal atoms. Web study with quizlet and.
SO2(Sulfur Dioxide) Molecular Geometry & Lewis Structure Geometry of
Which of the following constitutes a covalent. Web covalent radius half of the distance between two atoms within a single covalent bond. Is sulfur trioxide ionic or covalent? In fact, it is a multivalent atom, meaning it can make different number of bonds, up to 6, depending on the chemical formula. Is determined by the.
Molecules Free FullText Thiosulfoxide (Sulfane) Sulfur New
Web these four electrons can be gained by forming four covalent bonds, as illustrated here for carbon in ccl 4 (carbon tetrachloride) and silicon in sih 4 (silane). Web up to 6% cash back how many single covalent bonds would the element sulfur be expected to form in order to obey the octet rule? The.
Given below shows the bonding structure of sulphuric acid.Explain even
Values are given for typical oxidation number and coordination. Web up to 6% cash back how many single covalent bonds would the element sulfur be expected to form in order to obey the octet rule? It is the eighth most abundant element in the human body by weight, about equal in abundance to potassium, and.
Why can sulfur bond 6 times?
Web covalent radius half of the distance between two atoms within a single covalent bond. Web each of the 3 chlorines then forms a covalent bond by merging the atomic orbital containing its unpaired electron with one of the phosphorus's unpaired electrons. Which of the following constitutes a covalent. Web oxygen and other atoms in.
CH103 Chapter 5 Covalent Bonds and Introduction to Organic Molecules
Web each of the 3 chlorines then forms a covalent bond by merging the atomic orbital containing its unpaired electron with one of the phosphorus's unpaired electrons. The carbon atom is unique among elements in its tendency to form extensive networks of covalent. Group 5a form 3 bonds; In fact, it is a multivalent atom,.
Reading Covalent Bonds Biology I
Web how many single covalent bonds can halogens form? Web now sulfur has 6 unpaired electrons which means it can form 6 covalent bonds to give a total of 12 electrons around its valence shell. Web each of the 3 chlorines then forms a covalent bond by merging the atomic orbital containing its unpaired electron.
sulfur Definition, Element, Symbol, Uses, & Facts Britannica
Fluorine and the other halogens in group 7a (17) have seven valence electrons and can. The lewis structure for the sulfate ion consists of a central sulfur atom with four single bonds to oxygen atoms.â this yields the expected. Group 5a form 3 bonds; Because hydrogen only needs two electrons to fill its valence shell,.
SO2(Sulfur Dioxide) Lewis Structure, Hybridization, Molecular Geometry
A 70 kg (150 lb) human body contains about 140 grams of sulfur. It is the eighth most abundant element in the human body by weight, about equal in abundance to potassium, and slightly greater than sodium and chlorine. Is assigned as 2, since two hydrogens are required to satisfy bonding needs of these atoms..
How Many Covalent Bonds Can Sulfur Form Which of the following constitutes a covalent. The octet rule states that atoms of all elements forms bond with other atoms in such a way that each atom has eight electrons (octet) in its outermost. Web atoms of different elements will form either one, two, three or four covalent bonds with other atoms. Web each of the 3 chlorines then forms a covalent bond by merging the atomic orbital containing its unpaired electron with one of the phosphorus's unpaired electrons. It is the eighth most abundant element in the human body by weight, about equal in abundance to potassium, and slightly greater than sodium and chlorine.
The Octet Rule States That Atoms Of All Elements Forms Bond With Other Atoms In Such A Way That Each Atom Has Eight Electrons (Octet) In Its Outermost.
Finally, if both of sulfur's. Group 6a form 2 bonds; For example, the hydrogen molecule, h 2, contains a covalent bond. Web formation of covalent bonds.
Web If One Of Sulfur's Electron Pairs Is Separated, As Illustrated In The Middle Structure In Figure \(\Pageindex{1}\), Four Covalent Bonds Can Be Created.
Values are given for typical oxidation number and coordination. Because hydrogen only needs two electrons to fill its valence shell, it is an exception to the octet rule and only. Web atoms of different elements will form either one, two, three or four covalent bonds with other atoms. Web these four electrons can be gained by forming four covalent bonds, as illustrated here for carbon in ccl 4 (carbon tetrachloride) and silicon in sih 4 (silane).
How Many Covalent Bonds Can Each Carbon Atom Form?
In fact, it is a multivalent atom, meaning it can make different number of bonds, up to 6, depending on the chemical formula. It is the eighth most abundant element in the human body by weight, about equal in abundance to potassium, and slightly greater than sodium and chlorine. Is assigned as 2, since two hydrogens are required to satisfy bonding needs of these atoms. Nonmetal atoms frequently form covalent bonds with other nonmetal atoms.
The Potential Energy Of Two Separate Hydrogen Atoms (Right) Decreases As.
The lewis structure for the sulfate ion consists of a central sulfur atom with four single bonds to oxygen atoms.â this yields the expected. Web up to 6% cash back how many single covalent bonds would the element sulfur be expected to form in order to obey the octet rule? Fluorine and the other halogens in group 7a (17) have seven valence electrons and can. A 70 kg (150 lb) human body contains about 140 grams of sulfur.