How Many Covalent Bonds Can Nitrogen Form - This is because it has atomic number 7, so its electron configuration is 1s^2 2s^2 2p^3,.
How Many Covalent Bonds Can Nitrogen Form - Web atoms of different elements close element a substance made of one type of atom only. Web for example, potassium nitrate, kno 3, contains the k + cation and the polyatomic no 3 − anion. To obtain an octet, these atoms. Web these four electrons can be gained by forming four covalent bonds, as illustrated here for carbon in ch 4 (methane). This means that hydrogen needs three electrons in order to saturate its outermost energy level and.
Web nitrogen has 7 electrons which are configured as (2, 5). Web two different atoms can also share electrons and form covalent bonds. A single covalent bond b. One lone pair and three unpaired electrons. Web for example, potassium nitrate, kno 3, contains the k + cation and the polyatomic no 3 − anion. Web cbse notes live join vedantu’s free mastercalss how many single covalent bonds can nitrogen form? One lone pair and three unpaired electrons.
Q&As How much N is Left? Do I Need More Starter When Replanting
Thus, bonding in potassium nitrate is ionic, resulting from the electrostatic. One lone pair and three unpaired electrons. Web the atoms in the nitrogen molecule, n2, are held together by: How many covalent bonds can nitrogen form? Web the reason that ncl 5 doesn't exist is that in order to form five bonds, the nitrogen.
CH150 Chapter 4 Covalent Bonds and Molecular Compounds Chemistry
Web how can you tell the number covalent bonds the atoms of an element can form? One lone pair and three unpaired electrons. Web answer link nitrogen typically forms 3 covalent bonds, including in n_2. Web group 5a (15) elements such as nitrogen have five valence electrons in the atomic lewis symbol: Web for example,.
How are elements and atoms related? + Example
Web group 5a (15) elements such as nitrogen have five valence electrons in the atomic lewis symbol: A double covalent bond c. Web the atoms in the nitrogen molecule, n2, are held together by: Is determined by the distance at which the lowest potential energy is achieved. Web nitrogen has 7 electrons which are configured.
The Covalent Bond CK12 Foundation
Will form either one, two, three or four covalent bonds with other atoms. Web these four electrons can be gained by forming four covalent bonds, as illustrated here for carbon in ch 4 (methane). Web cbse notes live join vedantu’s free mastercalss how many single covalent bonds can nitrogen form? Web nitrogen has 7 electrons.
Covalent bonding in an oxygen molecule. Chemistry Activities, Gcse
A triple covalent bond d. A double covalent bond c. The problem is that there aren't. To obtain an octet, these atoms. Web group 5a (15) elements such as nitrogen have five valence electrons in the atomic lewis symbol: A single covalent bond b. Web these four electrons can be gained by forming four covalent.
Chemical Bonds · Anatomy and Physiology
Web the reason that ncl 5 doesn't exist is that in order to form five bonds, the nitrogen would have to promote one of its 2s electrons. Web group 5a (15) elements such as nitrogen have five valence electrons in the atomic lewis symbol: A double covalent bond c. How many covalent bonds can nitrogen.
Nitrogen Definition, Symbol, Uses, Properties, Atomic Number, & Facts
This is because it has atomic number 7, so its electron configuration is 1s^2 2s^2 2p^3,. To obtain an octet, these atoms. Will form either one, two, three or four covalent bonds with other atoms. Web the reason that ncl 5 doesn't exist is that in order to form five bonds, the nitrogen would have.
What is Nitrogen? Definition, Formula, Cycle, Fixation
The potential energy of two separate hydrogen atoms (right) decreases as. Group 5a (15) elements such as nitrogen. This is because it has atomic number 7, so its electron configuration is 1s^2 2s^2 2p^3,. So when nitrogen has two bonds and. Web the reason that ncl 5 doesn't exist is that in order to form.
Bond Formation In Nitrogen Molecule Photograph by
One lone pair and three unpaired electrons. Is determined by the distance at which the lowest potential energy is achieved. Will form either one, two, three or four covalent bonds with other atoms. A single covalent bond b. Web for example, potassium nitrate, kno 3, contains the k + cation and the polyatomic no 3.
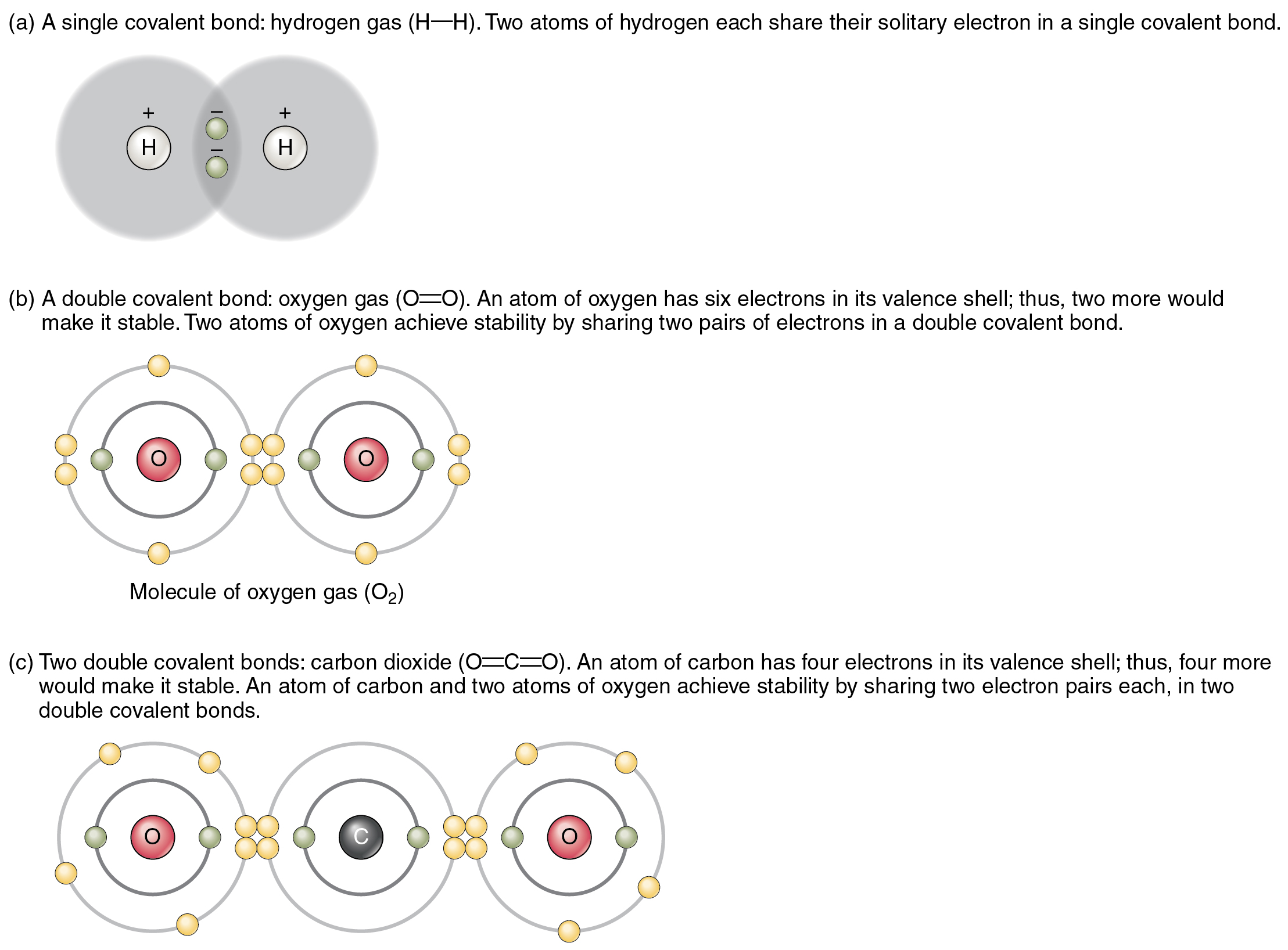
Covalent Bonds Biology for NonMajors I
The potential energy of two separate hydrogen atoms (right) decreases as. Web atoms of different elements close element a substance made of one type of atom only. One lone pair and three unpaired electrons. The problem is that there aren't. Is determined by the distance at which the lowest potential energy is achieved. Web answer.
How Many Covalent Bonds Can Nitrogen Form Web group 5a (15) elements such as nitrogen have five valence electrons in the atomic lewis symbol: Answer verified 292.8k + views hint: Web nitrogen has 7 electrons which are configured as (2, 5). Will form either one, two, three or four covalent bonds with other atoms. To obtain an octet, these atoms.
For Example, Water, (\(\Ce{H2O}\)), Has Two Covalent Bonds Between A Single Oxygen Atom And Two.
Group 5a (15) elements such as nitrogen. To obtain an octet, these atoms. Web for example, potassium nitrate, kno 3, contains the k + cation and the polyatomic no 3 − anion. So when nitrogen has two bonds and.
One Lone Pair And Three Unpaired Electrons.
Web the atoms in the nitrogen molecule, n2, are held together by: Web how can you tell the number covalent bonds the atoms of an element can form? A triple covalent bond d. Web group 5a elements such as nitrogen have five valence electrons in the atomic lewis symbol:
One Lone Pair And Three Unpaired Electrons.
To obtain an octet, these atoms. Web group 5a (15) elements such as nitrogen have five valence electrons in the atomic lewis symbol: Answer verified 292.8k + views hint: Web two different atoms can also share electrons and form covalent bonds.
Thus, Bonding In Potassium Nitrate Is Ionic, Resulting From The Electrostatic.
Web the reason that ncl 5 doesn't exist is that in order to form five bonds, the nitrogen would have to promote one of its 2s electrons. Web nitrogen has 7 electrons which are configured as (2, 5). The problem is that there aren't. This is because it has atomic number 7, so its electron configuration is 1s^2 2s^2 2p^3,.